With FDA clearance for spine surgery, SyncAR Spine brings patient MRI data directly into the operating room, advancing precision and supporting less invasive techniques
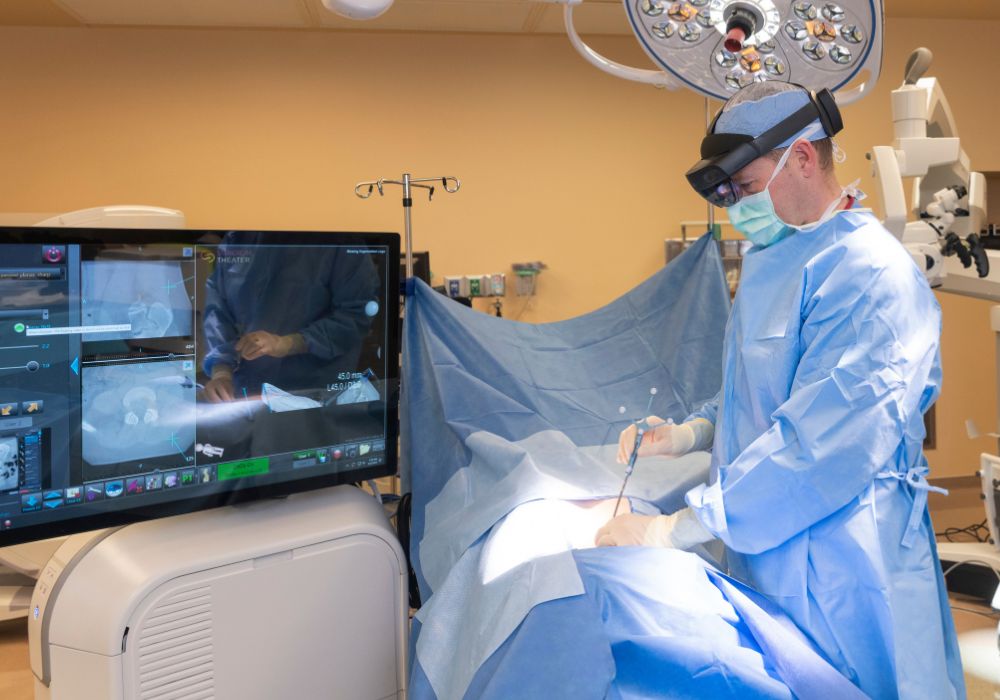
INDIANAPOLIS—Dr. Gregory Poulter, a nationally recognized spine surgeon at Indianapolis-based OrthoIndy, is the first surgeon worldwide to perform a SyncAR® Spine case using Surgical Theater’s next-generation spine platform.
The U.S. Food and Drug Administration (FDA) recently granted 510(k) clearance for SyncAR Spine, expanding the platform’s capabilities with advanced XR tools, such as augmented and virtual reality, powered by AI algorithms. This means MRI is now available alongside CT in the operating room, giving surgeons greater clarity and control in spine surgery by helping align preoperative planning with intraoperative navigation. Dr. Poulter says having MRI data overlaid in his field of view has changed the way he approaches procedures.
“Spine surgery often involves trimming bones to unpinch nerves, and the standard way of doing surgery meant removing bone without being able to see what was underneath,” said Poulter. “Now, we can bring MRI into our field of view and visualize nerves and discs in real-time. This level of detail allows us to be more precise and less invasive, all to the benefit of our patients.”
Integrating MRI data is just one way that SyncAR spine builds on its proven foundation in operating rooms nationwide. The platform transforms traditional imaging into a dynamic surgical roadmap, fully integrating with Medtronic StealthStation™ navigation and featuring:
- AI-driven vertebra segmentation to streamline planning
- Advanced Decompression Planning (ADP) to pre-map resections before surgery
- Segmental Fusion to keep preoperative models aligned with intraoperative CT scans, even as patient positioning changes
- Voxel-level drill tracking to monitor bone removal in real-time
“Seeing MRI data live in the OR, aligned seamlessly with navigation, is the greatest improvement in navigation since its inception nearly two decades ago,” added Poulter. “I can plan every detail in advance and then see that plan projected in real time during surgery, fully aligned with my workflow.”
The FDA clearance marks a major evolution of Surgical Theater’s eXperiential Reality (XR) platform, validated through comprehensive testing and AI-model performance studies. It reinforces the company’s leadership in surgical XR and sets a new standard for intraoperative visualization.
Unlike platforms that rely on headset-only overlays or proprietary navigation, SyncAR Spine was designed as a comprehensive surgical XR platform. It integrates seamlessly with existing workflows – enhancing navigation rather than replacing it – and provides richer intraoperative visualization without disrupting routines.
With more than 50,000 clinical uses within its XR platforms, Surgical Theater continues to set the standard in surgical visualization. Alon Zuckerman, president at Surgical Theater, noted, “SyncAR Spine represents the next step in that journey, bringing surgeons deeper insight, stronger alignment between plan and execution and the unprecedented ability to carry MRI into the OR in real time. This is a transformational leap in spine surgery.”
For more information about SyncAR Spine, visit surgicaltheater.com. To learn more about OrthoIndy and its team of spine specialists, visit OrthoIndy.com.